Planova™ BioEX Virus Removal Filters
High Filtration Performance
and Robust Virus Removal Capability
Planova™ BioEX filters deliver high filtration performance even for biopharmaceuticals that contain proteins known to cause clogging. It has a proven track record in virus filtration for a wide range of products and delivers high performance even under high concentration conditions.
- High filtration performance for high concentration solution conditions
- Reduced filtration time enabled by the high pressure resistance of the PVDF membrane
- Robust virus removal performance unaffected by pH or temperature
Virus Removal Filter - Planova™ Lineup
Features
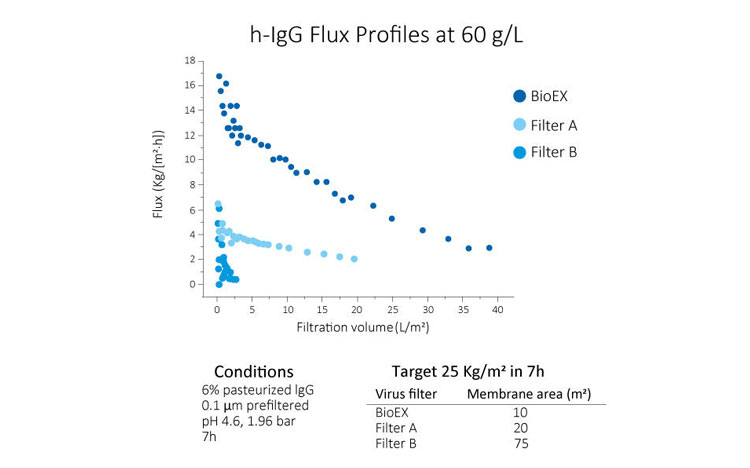
High Filtration Performance for High Concentration Solution Conditions
Planova BioEX delivers exceptional filtration performance for formulations that are considered difficult to filter, enabling smooth process development.
Even for high concentration formulations and molecules that are challenging to filter with other filters, Planova BioEX maintains high filtration performance.
Even for high concentration formulations and molecules that are challenging to filter with other filters, Planova BioEX maintains high filtration performance.
Reduced Filtration Time Enabled by the High Pressure Resistance of the PVDF Membrane
The unique hydrophilized polyvinylidene fluoride (PVDF) membrane of Planova BioEX provides outstanding pressure resistance, allowing processing at high flux. This capability helps shorten virus filtration time, typically finishing within 4 to 8 hours in a single manufacturing shift.
Even under demanding conditions—such as high pressure, high concentration, or large-volume filtration—Planova BioEX maintains stable flux with minimal decline. This consistent performance ensures safe and efficient virus filtration.
Even under demanding conditions—such as high pressure, high concentration, or large-volume filtration—Planova BioEX maintains stable flux with minimal decline. This consistent performance ensures safe and efficient virus filtration.
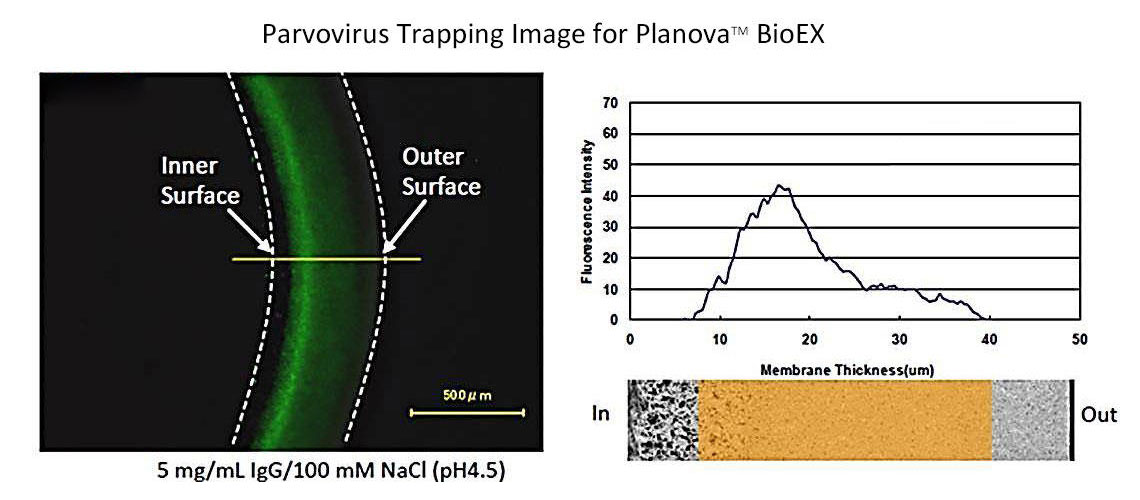
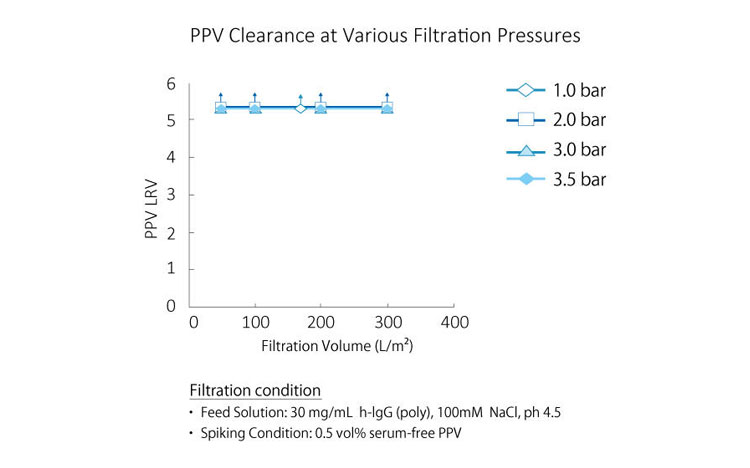
Robust Virus Removal Performance Unaffected by pH or Temperature
Planova BioEX features a thick membrane structure that captures viruses across a large membrane area, ensuring robust virus removal.
As shown in the graph below, Planova BioEX delivers consistent virus removal performance under various conditions, including different buffer compositions and filtration pressures.
As shown in the graph below, Planova BioEX delivers consistent virus removal performance under various conditions, including different buffer compositions and filtration pressures.
| pH | Conductivity (mS/cm) |
Pool MVM LRV | ||
|---|---|---|---|---|
| mAb 2 | mAb 1 | mAb 3 | ||
| pI 7.2 | pI 8.6 | pI 9.0 | ||
| 4 | 20 | ≥5.02 | ≥5.52 | 5.45 |
| 8 | 20 | ≥5.53 | ≥5.53 | ≥5.62 |
| 4 | 3 | ≥5.48 | ≥5.07 | ≥5.02 |
| 8 | 3 | ≥5.33 | ≥5.40 | ≥4.87 |
| 5.5 | 7 | ≥5.07 | ≥4.87 | ≥5.20 |
Adapted from: Joshua Goldstein, Janssen, 2014 Planova Workshop
Documents
Spotlights
Download technical leaflets highlighting the key features of Planova BioEX filters.
Each Spotlight presents customer and internal data on a subset of features.
Each Spotlight presents customer and internal data on a subset of features.
Planova Spotlight Vol.5[PDF:1.4MB]
Enable continuous processing with robust viral clearance
Planova BioEX Spotlight Vol.3[PDF:1.2MB]
Ensure smooth scale-up with consistent performance, high recovery, and easy operation
Planova BioEX Vol. 2[PDF:2.7MB]
Accelerate early development with consistent flux and robust virus removal you can trust
Planova BioEX Spotlight Vol. 1[PDF:2.8MB]
Discover reliable filterability, robust virus removal capacity and proven scalability
Technical Documents
Access technical documents for Planova BioEX filters through the links below.
Related Presentations
Access presentations on Planova BioEX filters through the links below.
Related Videos
Access videos on Planova BioEX filters through the links below.

BioEX Lab-scale Operation Guide

Planova BioEX: Leading the Next Generation of Virus FIlters

Minimizing Resources in Developing Virus Filtration Process with Planova BioEX

Manufacturing Considerations for Virus Filtration Processes Using Planova
Related Publications
Access publications on Planova BioEX filters through the links below.
- Adapting virus filtration to continuous processing: Effects of product and process variability on filtration performance
- Effect of pH, NaCl concentration, and mAb concentration of feed solution on the filterability of Planova™ 20N and Planova™ BioEX
- Development of a Small-Scale Continuous Virus Filtration Model That Incorporates Inline Spiking and Mixing
Product Specifications
| Planova BioEX | ||||||||
|---|---|---|---|---|---|---|---|---|
| Effective surface area (m²) | 4.0 | 1.0 | 0.1 | 0.01 | 0.001 | 0.0003 | ||
| Component | Hollow fiber membrane | Hydrophilic modified polyvinylidene fluoride | ||||||
| Housing and headers | Polycarbonate | |||||||
| Sealant | Polyurethane | |||||||
| O-rings | Silicone | |||||||
| Nozzle plugs | Silicone | - a | ||||||
| Nozzle caps | - | Silicone | ||||||
| Ferrule caps | Polycarbonate | - | ||||||
| Luer lock plugs | - | Polycarbonate | ||||||
| Balloon cap holders | Polycarbonate | - | ||||||
| Gaskets b | Silicone | - | ||||||
| Balloon caps | Silicone | - | ||||||
| Nozzle stoppers | - | Silicone | - | |||||
| Clamp bands | Polysulfone | - | ||||||
| Threaded clamps | Clamp bolts | - | Polypropylene | - | ||||
| Clamp nuts | - | Polypropylene | - | |||||
| Locknuts | Polycarbonate | - | ||||||
| Supplied as | Filled with purifled water c | |||||||
| Sterilization method | Autoclaving | |||||||
| Packaging format | Packed individually in sterilization bags | |||||||
| Operating pressure (TMP) | ≤343 kPa | |||||||
| Operating pH | 2-9 | |||||||
a Not applicable
b Gaskets are used for connecting quick clamps to nozzles of Planova BioEX filters with effective surface areas of 4.0, 1.0 and 0.1 m2.
c Purified water in 4.0, 1.0 and 0.1 m2 Planova BioEX filters contains <0.1% NaCl.
b Gaskets are used for connecting quick clamps to nozzles of Planova BioEX filters with effective surface areas of 4.0, 1.0 and 0.1 m2.
c Purified water in 4.0, 1.0 and 0.1 m2 Planova BioEX filters contains <0.1% NaCl.
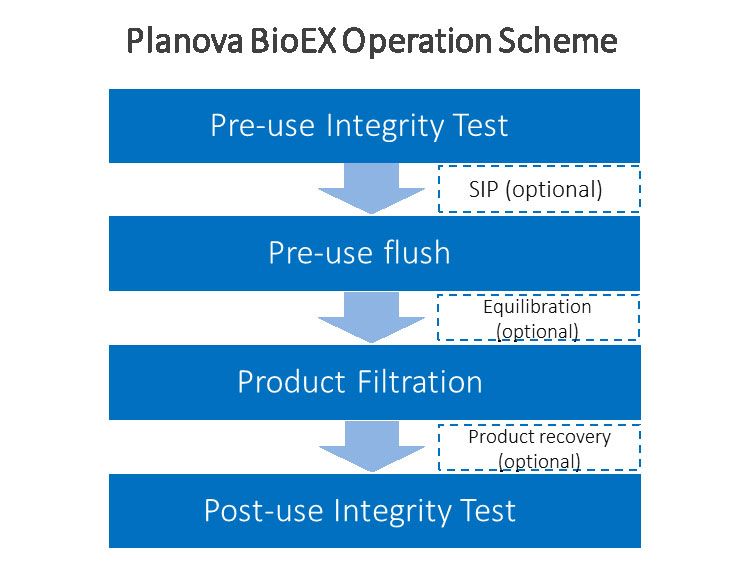
Integrity Testing
Integrity testing of Planova BioEX filters is performed to confirm the integrity of filters used during filtration.
Because the hydrophilized polyvinylidene fluoride (PVDF) membrane used in Planova BioEX offers high mechanical strength, the integrity test is simplified.
The leakage test is conducted pre-use and post-use to confirm that the filter is free from gross defects such as pinholes and other membrane damage.
The leakage test is conducted pre-use and post-use to confirm that the filter is free from gross defects such as pinholes and other membrane damage.
- Pre-use: Detect defects that may have occurred during shipping
- Post-use: Detect defects that may have developed during filtration
Related Products & Services
Related Products
Services
Ordering Information
Please reference the catalog number when placing orders.
| Effective surface area (m²) | Catalog number | |
|---|---|---|
| Planova BioEX | 4.0 | EX4-0000 |
| 1.0 | EX1-0000 | |
| 0.1 | EXZ-1000 | |
| 0.01 | EXZ-0100 | |
| 0.001 | EXZ-0010 | |
| 0.0003 | EXZ-0003 |














.png)